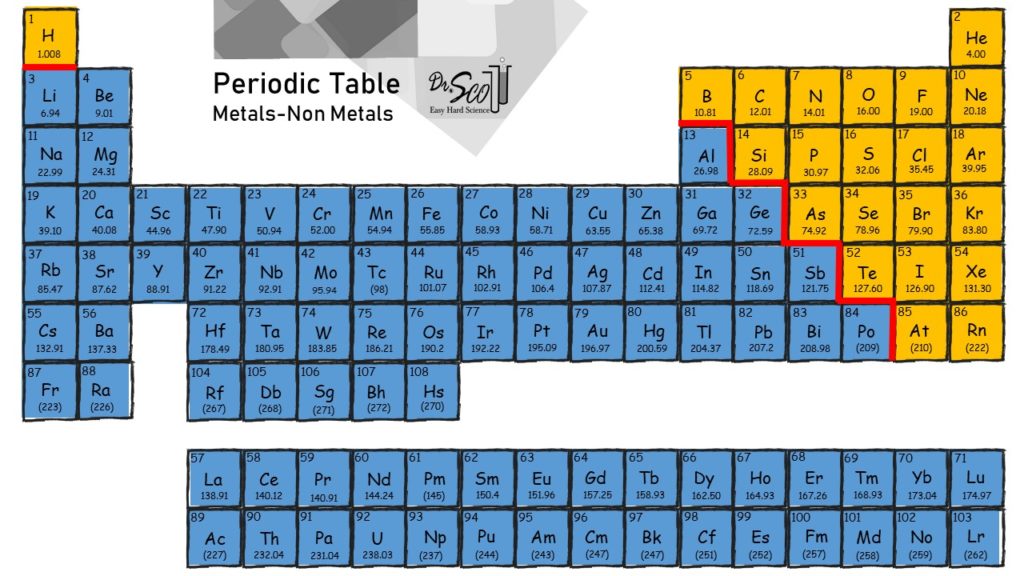
Examples of elements include oxygen (O), carbon (C), and gold (Au).Ī compound is a substance that is made up of two or more elements and whose composition is constant. The periodic table of the elements lists all of the discovered elements and their atomic numbers. This is how we identify one element from another. We cannot use chemical methods to break hydrogen down into anything simpler.Įach element has a unique atomic number, which is the number of protons it has. For example, the element hydrogen has one proton, one neutron, and one electron. An element consists of an atom with a unique combination of protons, neutrons, and electrons. compoundĪn element is a substance that cannot be separated into simpler substances by chemical means. In this article, we will define the terms element and compound and explain how they are used differently in chemistry. If you need a simple explanation of what these terms mean, we have your solution. The terms element and compound are commonly used in chemistry. A compound is a substance made of two or more elements chemically combined and with a consistent composition, such as hydrogen and oxygen combining into water (H ₂ O). The periodic table of elements lists all discovered elements. The "-ic" suffix is used for the acid that has more oxygen atoms.An element is a substance that cannot be further broken down into smaller substances by chemical means, such as hydrogen (H). Oxoacids containing oxygen use the "-ous" or the "-ic" suffix. Hydro acids use the prefix "hydro-" and the suffix "-ic". There are a few exceptions that end in "-ide" including hydroxide, peroxide, and cyanide. Polyatomic compounds use a different suffix. Using the stock method, a roman numeral is used after the metal to indicate which ion is using the charge. If one of the two compounds is a metal, then the naming convention changes a bit. See below for some of the more complex naming rules. In a compound that contains carbon and oxygen the element to the left on the periodic table (carbon) would go first.In a compound that contains iron and fluoride, the metal (iron) would go first.If there are two nonmetal elements, then the first name is the element to the left side of the periodic table. If the compound is made of a metal element and a nonmetal element, then the metal element is first. When there are two elements in a compound, which element goes first in the name? How is the order of the elements determined? ** note: the "mono" prefix is not used on the first element. In cases where there is more than one atom (for example there are two oxygen atoms in CO 2) you add a prefix to the start of the element based on the number of atoms. 
To get the second word we use the name of the second element and change the suffix to "ide" at the end of the word. To get the first word we use the name of the first element, or the element to the left of the formula. The name of a compound with two elements has two words. The name is built from the elements and the construction of the molecule.įirst we'll cover how to name molecules with two elements (binary compounds). It is a standard method of naming compounds that is used by scientists around the world. You can go here to learn more about molecules and compounds.Ĭhemists have a specific way of naming compounds. A compound is a type of molecule with more than one element. Compounds have their own properties that are unique from the elements they are made of. These bonds are so strong that the compound behaves like a single substance. Chemical compounds are formed when elements are joined by chemical bonds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
